"is this a wake or revival?" Jim Brown
March 21-22, 2012, Santa Barbara, CA. National Center for Ecological Analysis and Synthesis (NCEAS) symposium.
A special invitation- only symposium marking the end of NCEAS as we know it, saw a number of interesting talks and retrospectives about where NCEAS has been and where it is going. 170 people attended, including some former postdocs, working group participants and leaders in ecology. The reason for this introspective meeting is that NCEAS's core NSF funding is about to end, without renewel. Jim Brown's quote from his talk, whether we were here for a wake or a revival really captured the spirit of the meeting.
The goals were twofold. First was to look back and celebrate the accomplishments of NCEAS. University of California at Santa Barbara is globally one the top influential research institutions in the world, and this success has been driven in large part by the success of NCEAS. More than 5000 people have come to NCEAS and their efforts have resulted in thousands of publications, and many citation classics. The early visions of NCEAS were broad and fuzzy and by all accounts NCEAS has exceeded all expectations.
The second motivation for ts meeting was to think about the future. What can NCEAS be under different funding regimes, and how should it move forward? The is no doubt that it will be fundamentally different, but can there be a successful continuation of the NCEAS model, will it die, or will it give birth to a new enerprise, NCEAS 2.0?
The symposium saw great talks, from people like Jim Brown and Jane Lubchenco, and interesting panel discussions on numerous topics (see #treas2012 in twitter for synopsis of the meeting). There were a lot of past tense statements.
However, it was clear that there was much to celebrate. NCEAS clearly impacted ecology. Did its success simply coincide with cultural changes in the field or did it drive changes? The consensus was that it drove changes. It fostered large collaborations. Dave Tilman said that before NCEAS, ecology was largely local and lab-driven, but NCEAS offered a way to get people together to ask bigger questions. The postdoctoral fellows have been extremely successful, with the vast majority ending up in faculty positions in top institutions. It was acknowledged that many sub fields were created or coalesced at NCEAS, including disease ecology and metacommunity dynamics.
Why has it been so successful? NCEAS is a special inclusive place where people want to come, away from their responsibilities. The technical help here and expertise that made anything possible, any data challenges were overcome and analytical difficulties solved. Postdocs were given complete independence and were allowed to pursue collaboration and networking. Jim Brown remarked that NCEAS is the single greatest event in the history of ecology. Subfields now talk, lab projects are now geared towards collaboration and linkages with other work in ways that did not exist before.
So then, what will the future hold for NCEAS? The answer to this was left vague and uncertain. People argued for what NCEAS 2.0 should look like. For example, it was argued that NCEAS 2.0 should resurface something like science 2.0, making the focus data and data sharing, changing methods and philosophy of how science is done. Massive anonymous collaboration requires assumed standards and altruism. Other arguments focused on the need for NCEAS to reach out to new partners and to go global.
Peter Karieva said it well. NCEAS 2.0 should be interacting with major corporations, since they represent the drastic impacts on ecological systems around the world. 1.0 was about data accessability, 2.0 should about applicability and tools to affect change.
Whatever NCEAS 2.0 looks like, it will be different. There seems to be two ways forward. One is that it struggles to maintain its past activities or one that like the Phoenix rises from the ashes and boldly goes forward to again push the ecology in new directions.
Thursday, March 22, 2012
Sunday, March 11, 2012
On rejection: or, life in academia
I guess it’s not surprising, given that I’ve written about
failure in science, that I would write a post about rejection as well.
Actually, I’m not so interested in writing about rejection as I am in hearing
how people have learned to deal with it.
Academia is a strange workplace. It’s stocked with bright
people who’ve been successful throughout their previous academic
endeavours (with some exceptions*). For the most part, they haven’t faced too
much criticism of their intellectual abilities. But in academia you will spend
your career being questioned and criticized, in large part by your peers. You
will constantly be judged (with every submitted manuscript, grant application,
or tenure review). And this is the universal truth about academia: you will be
rejected. And for some (many?) people, that's a difficult thing to accept.
Rejection may be so painful in part because it can be hard
to interpret. After all, it’s an old trope that rejection is a normal part of
academia. But how much rejection is normal, when is it just a numbers game and when
is it a sign of professional failing? Let alone the fact that rejection depends
on a shifting academic landscape where available funding, journal quotas, and
research caliber are always changing. So I’m curious: does the ability to deal
with rejection factor into academic success? Are some people, based on
personality, more likely to weather rejections successfully, and does
this translate into academic success? Or is the development of a thick skin
just the inevitable outcome of an academic life?
*A couple of the people I know who are generally unfazed by
rejections would say that they deal well with rejection because they weren’t
particularly great students and so academic failure isn’t new or frightening to
them.
Friday, March 2, 2012
The niche as a changeable entity: phenotypic plasticity in community ecology
Nearly all explanations for coexistence in communities focus
on differences between species. The scale of these differences may occur over
large temporal (e.g. evolutionary history, phylogenetic relationships) or
spatial scales (e.g. environmental tolerances), or at the scale of the
individual. In plants, interactions at the local scale are given particular
attention, including interactions mediated by trait differences between species.
At finer scales still, there has been recent focus on differences between
individuals of the same species, whether they are driven by genotypic
differences (link) or plastic changes in individual phenotypes.
 |
| From Ashton et al. 2010 |
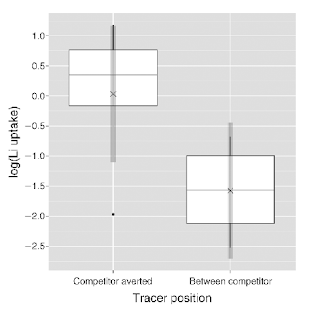 |
| From Schiffer et al. 2011, Lithium uptake is significantly higher on the non-competitor side |
A couple of papers from the last few years provide tantalizing glimpses into the possible contribution of plasticity to coexistence. In Schiffers et al. (2011), the authors use experimental and modeling approaches to test whether root uptake can change in response to the proximity of competitors. In the experimental study, the authors looked at the uptake of lithium (a stable nutrient that will be taken up in the place of potassium) by Bromus hordeaceus. They planted pairs of B. hordeaceus at varying distances apart and then injected lithium into the soil at different differences from the focal plant. They found that lithium uptake was significantly higher on the non-competitor side of the focal plant than on the competitor side, suggesting that plastic changes in resource uptake occurred in response to competitor proximity. Modelling results from the same study suggest that plasticity may allow individuals minimize competitive pressure by making changes in belowground architecture, thereby using available space more efficiently.
Ashton et al. (2010) take a similar approach, looking at how
the uptake of nutrients (in this case three forms of nitrogen (N)) varies among
species depending on their competitive environment. They explored the ways in
which plasticity could lead to changes in the realized niche. In particular,
they explored two hypotheses: that plants would exhibit niche preemption, where
the inferior competitor switched to a different form of nitrogen in the
presence of the superior competitor; or dominant plasticity, where plasticity
actually enhances competitive ability.
The authors looked at 4 species, 3 common and 1 rare(r), in an alpine tundra
community, isolating naturally occurring pairs of each combination of species.
These ‘competitive arenas’ were isolated, and other species within the arena
were removed. After a year, the authors added N15 tracers to each
arena, in three forms (NH4+, NO3-, and glycine): these tracers would allow them
to track the N once it was incorporated into the plant tissue. The plants were
then harvested and the amount of each type of nitrogen in each was measured.
Plant biomass was also recorded, and used to estimate the ‘competitive
response’ (basically the ratio of biomass when grown with a competitor compared
biomass to when grown solo). Their findings were rather neat: the 3 common
plants experienced no negative effect on biomass from growing in competition with
the rare plant, but the rare plant had much lower biomass when grown in the
presence of any of the common plants. Further, while the common plants showed
changes in the form of N they used when growing with the rare plant, the rare
plant did not switch its N preference. The rare plant’s lack of plasticity in
response to competition may relate to its lower biomass when grown with
superior competitors, and ultimately its lower abundance.
Although limited, these studies hint at the role that
phenotypic plasticity could play in interspecific interactions. Unfortunately
plasticity may be difficult to measure in many contexts, particularly since
variation within a species can be attributed to genetic differences or
phenotypic plasticity, and these factors must be teased apart. Further, there
is an issue of differentiating the effects of resource limitations from ‘adaptive’
plastic changes in growth. While plants are relatively tractable for these
types of studies (they’re sessile, they use limited abiotic resources), other
organisms are less explored for a reason.
What these studies can’t address is the question of ‘how
important is phenotypic plasticity, really’? Reviews of coexistence mechanisms
list numerous possible ways by which coexistence is facilitated among species.
For plants especially, the limited number of resources required for survival
has lead to great consideration of the possible niche axes over which species
can differentiate themselves. Phenotypic plasticity's contribution to coexistence may be that it provides another way by which plants can partition resources at very fine scales. And if
nothing else, such results provide further evidence that variation within species
may be an important component of coexistence.
Thanks to Kelly Carscadden for discussions on the topic.
Subscribe to:
Posts (Atom)

